Prohibition Order Granted Against Generic VIREAD Product
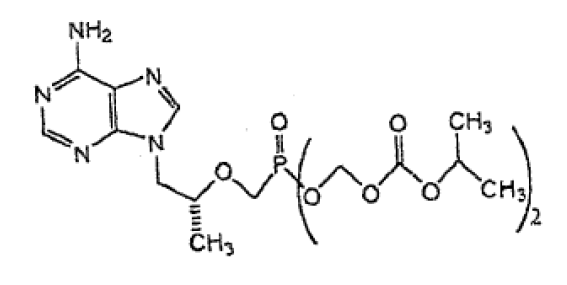 On August 23, 2016, the Federal Court released the public judgment and reasons by Justice Brown in 2016 FC 857. The case was a prohibition proceeding pursuant to s. 6 of the NOC Regulations relating to Canadian Patent No. 2,261,619, which expires on July 25, 2017. The 619 Patent claims tenofovir disoproxil fumarate – a prodrug present in Gilead’s VIREAD used in the treatment and prophylaxis of HIV. The prodrug increases the oral bioavailability of the parent drug, tenofovir. A separate companion decision of Justice Brown relating to Canadian Patent No. 2,512,475, listed against Gilead’s TRUVADA (tenofovir disoproxil fumarate + emtricitabine) product, is reported here.
On August 23, 2016, the Federal Court released the public judgment and reasons by Justice Brown in 2016 FC 857. The case was a prohibition proceeding pursuant to s. 6 of the NOC Regulations relating to Canadian Patent No. 2,261,619, which expires on July 25, 2017. The 619 Patent claims tenofovir disoproxil fumarate – a prodrug present in Gilead’s VIREAD used in the treatment and prophylaxis of HIV. The prodrug increases the oral bioavailability of the parent drug, tenofovir. A separate companion decision of Justice Brown relating to Canadian Patent No. 2,512,475, listed against Gilead’s TRUVADA (tenofovir disoproxil fumarate + emtricitabine) product, is reported here.
The only asserted claim of the 619 Patent was claim 32, directed to tenofovir disoproxil and its salts, tautomers and solvates. Justice Brown concluded that Apotex’s allegations of anticipation, obviousness, invalid selection patent, and lack of utility were not justified and issued the prohibition order sought by Gilead. Justice Brown also held that a previous decision on the 619 Patent involving a different generic was not binding on conclusions of invalidity.
Blinding of Experts
The parties chose different methods to gather information from their expert witnesses. Counsel for Gilead provided the legal framework to its experts early, including legal tests for anticipation, obviousness, and utility. Counsel for Apotex did so after the experts had drawn their own conclusions on issues such as the promise of the patent, claim construction, and the prior art, thereby “blinding” its experts.
Justice Brown concluded that the blinding of a witness is one of perhaps several factors that goes to the weight of evidence, but it is not a matter that goes to admissibility. He held that the blinding issue is a question of relevance, reliability and weight, and is not a doctrinal matter. Generally, Justice Brown preferred the expert testimony of Gilead’s expert witnesses in part because of the greater experience and knowledge of Gilead’s expert witnessnes compared to Apotex’s expert witnesses. One of Apotex’s witnesses also admitted that his affidavit contained two significant false statements regarding the state of the art.
Anticipation
Justice Brown concluded that claim 32 of the 619 Patent was not anticipated by EP 0 481 214 because the latter did not disclose tenofovir disoproxil or even that a prodrug moiety could contain a carbonate group. Justice Brown deemed it significant that Gilead’s expert witness explained that the skilled person would not have understood that EP 214 included carbonates, because of the conspicuous absence of carbonates from any explicit disclosure, including the complete absence of carbonates from the explanations and examples provided in EP 214.
Obviousness
Justice Brown concluded that tenofovir disoproxil was not obvious or obvious to try. Justice Brown, reading the 619 Patent through the eyes of the unimaginative skilled person, was unable to find that the carbonate subgroup was uninventive and either self-evident or “obvious to try”. The prodrug literature on phosphonate nucleotides for antivirals suggested that carbonates would be unstable. Justice Brown further held that Gilead’s research team at that time was acting with an inventive spark and expected a period of trial and error before achieving their goal.
Selection Patent
Justice Brown did not accept Apotex’s allegation that claim 32 of the 619 Patent was an invalid selection from the genus disclosed in EP 214. He held that even if EP 214 included tenofovir disoproxil (which he found that it did not) Gilead’s prodrug presented special advantages for efficient oral delivery that were not previously disclosed. He noted Examples 15 and 16 of the 619 Patent regarding the specific oral bioavailability of tenofovir disoproxil.
Inutility
Justice Brown concluded that the promise of the 619 Patent is efficient oral delivery of the parent compound (e.g. tenofovir) through its prodrug, which has a specific carbonate moiety, yielding tenofovir disoproxil. To establish utility, he stated that he is not bound only by the contents of the 619 Patent. He considered the tests disclosed in the 619 Patent and prior art disclosures, which showed improved bioavailability of tenofovir disoproxil over tenofovir.
Justice Brown granted Gilead’s prohibition application and awarded costs to Gilead.
A copy of Justice Brown’s decision may be found here.

