Clean Sweep for Teva in VELCADE Patent Action
In a Judgment dated July 18, 2018, Justice Locke allowed Teva’s claim for section 8 damages in relation to its bortezomib product, and dismissed Millennium’s counterclaim for the infringement of three patents, holding the asserted claims of two patents as invalid, and the asserted claims of a third patent not infringed.
As the parties had already reached agreement regarding matters of liability and quantification of damages, the trial only concerned Millennium’s claim for infringement of three patents and Teva’s defences based on non-infringement and invalidity.
Patent 2,203,936 (compound)
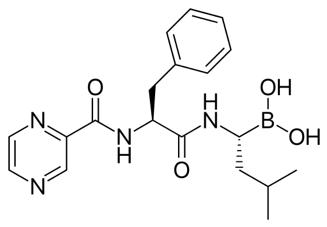
Claim 37 covered 6 boronic acid peptide analogs including bortezomib, whereas Claim 69 covered the compound (N-(2-pyrazine)carbonyl-L-phenylalanine-L-leucine boronic acid), also known as bortezomib, having the following structure:
Skilled Person: The parties had agreed that the relevant person skilled in the art was a team comprising a medicinal chemist with experience in boronic acid chemistry and a biochemist having experience with the proteasome and peptide chemistry. The common general knowledge included synthesis of N-terminal protected peptidyl boronic ester and acid compounds, and their effectiveness at inhibiting proteases.
Justice Locke accepted that WO 91/13904 describes a broad genus peptidyl compounds disclosed to inhibit proteases, including the proteasome. Justice Locke also ejected Millennium’s arguments that he 904 Application did not disclose the N-terminal blocking group of bortezomib, holding:
[169] … the broad range of possible N-terminal blocking groups that are expressly contemplated, together with the reference to known equivalents, leaves room for pyrazine carbonyl, provided it can be shown to be “known to protect molecules from degradation by aminopeptidases
Rather, Justice Locke held that while pyrazine carbonyl was an unusual choice, multiple references in the prior art established that it was a known N-terminal blocking group such that bortezomib falls within the broad genus of compounds contemplated in the 904 Application, and therefore the 936 Patent is effectively a selection patent and must meet the requirements for validity thereof.
Obviousness: Despite falling within the scope of compounds contemplated in the 904 Application, bortezomib was not specifically disclosed in the prior art. Millennium argued that bortezomib could not be obvious because a skilled person could not predict that bortezomib and the other claimed compounds would be effective inhibitors of the proteasome. Justice Locke rejected this argument holding nothing in the combination of the components of bortezomib prompts a conclusion that the selection of all of them together was any less obvious.
Further, the selection of bortezomib was not in respect of a “quality of a special character peculiar to the selected group”, as the compounds of the asserted claims did not contain anything would have been found by the unimaginative skilled technician without the exercise of inventive ingenuity.
Justice Locke also noted held that commercial success could not save the claims from obviousness because the 936 Patent was not sufficient to create a commercially successful product.
Patent 2,436,146 (formulation)
Justice Locke determined that claims 30, 45, 46 and 81-84 of the 146, generally directed to the lyophilized bortezomib mannitol ester, and methods for making it, were obvious.
Justice Locke accepted Teva’s argument that the skilled person is not only a pharmaceutical formulator, but is a team also containing a medicinal chemist. Boronic acid and ester compounds, including bortezomib, were known as part of the common general knowledge.
The parties agreed that the skilled person would be aware that lyophilizing a solution of bortezomib and mannitol could result in the formation of an ester. However, Millennium’s expert, Dr. Anderson, contended that the potential for ester formation would make the use of mannitol a non-starter because it would not be inert (a desired characteristic of a bulking agent). Justice Locke accepted the evidence of Teva’s expert, Dr. Bachovchin, that a skilled person would expect that any ester formation would be readily reversible by hydrolysis.
Justice Locke determined that the inventive concept of claim 30 is lyophilized bortezomib mannitol ester with its improved stability, rejecting Millennium’s argument that the inventive concept also includes other properties associated with the compound, including solubility and dissolution rate. This distinction was not critical, as Justice Locke found the claim to be obvious, even assuming the inventive concept included solubility and dissolution rate.
As mannitol had been used in the formation of an ester of another boronic acid, Justice Locke determined that the difference between the inventive concept and the state of the art was the use of mannitol and lyophilisation to form an ester of bortezomib.. In determining that it was more or less self-evident to try to form lyophilized bortezomib mannitol ester, Justice Locke concluded that it was more or less self-evident that the lyophilized bortezomib mannitol ester ought to work, holding:
[263] As indicated in paragraph [242] above, it was well-known to use lyophilisation to address aqueous instability. Of course, this is a general statement that does not take into account the other known possibility that lyophilizing a solution of bortezomib and mannitol could result in the formation of an ester (or several different esters). This possibility would introduce an additional uncertainty, but as indicated above, the skilled person would expect such ester formation to be readily reversible. The skilled person would also still expect improved stability as a result of lyophilisation.
[264] In my view, it was indeed more or less self-evident that what was being tried ought to work. It was not certain to succeed, but skilled person would have considered the probability good.
Justice Locke also held that the inventors’ actual course of conduct that was disclosed in the prior art was less helpful in determining whether the solution provided by the inventors was obvious to try at the claim date, holding that the skilled person would have had more information than the inventors, and would not need to go through all of the same steps that the inventors did.
Furthermore, the inventors’ initial search was found lacking, as it failed to identify prior art that was clearly relevant. An inventor, Dr. Stella, thought about trying lyophilization relatively early in his work, but the project manager initially opposed this approach. Once Dr. Stella began working on lyophilisation, he had the solution described in the patent within a month or two, and improved stability was verified with routine testing.
Patent No. 2,738,706 (synthetic method)
Justice Locke found that Teva’s processes did not infringe the 706 Patent, which is generally directed to a large-scale process for making bortezomib. At trial, Millennium had argued that several claim elements were not essential and therefore need not be in the Teva process in order for the Court to find infringement.
Justice Locke rejected Millennium’s proposed construction, holding that in purposively construing a patent, the language of the claims is prime. More specifically, Millennium had argued that a definition of “large scale” in the 706 Patent as requiring at least 5 moles of a starting reagent did not apply to the claims because it was directed to a different invention claimed in a separate divisional application. Justice Locke noted that this construction was not supported by any expert evidence, and held that the other passages in the disclosure do not detract or alter the explicit definition of “large scale”, holding:
[307] In my view, the passages cited above from paragraphs 8 and 25 of the 706 Patent do not detract from or alter the definition provided in paragraph 31. Those passages essentially serve to emphasize that there is no specific upper limit to the scale contemplated in the 706 Patent. On the other hand, the definition in paragraph 31 provides a lower limit to the scale. That definition applies to the term “large-scale”.
Justice Locke also held that the specific solvents recited in step (aa) of asserted claim 1 were “clear and unambiguous” and were essential elements that could not be substituted for other solvents. Justice Locke further held that the solvents employed by Teva work in a substantially different way than the solvents claimed in the 706 Patent, such that Teva did not infringe the 706 Patent.
Justice Locke similarly rejected Millennium’s arguments that various elements of steps (bb) and (cc) were not essential, explaining as follows:
[320] …As with step (aa), I can decide the issue of essentiality based on whether the skilled reader would have understood from the language of the claim that the patentee intended that strict compliance with the primary meaning was an essential requirement of the invention. As in step (aa), I see no ambiguity in the terms used in step (bb) that would lead to a conclusion that they encompass a range of alternatives.
In view of his findings of non-infringement, Justice Locke found it unnecessary to address the validity of the 706 Patent.
A copy of Justice Locke’s public Judgment and Reasons may be found here.
Teva was successfully represented by Aitken Klee’s David Aitken, Marcus Klee, Bryan Norrie and Jonathan Giraldi.